Seznamy 69 Geometry Of Central Atom Vynikající
Seznamy 69 Geometry Of Central Atom Vynikající. In this case the theoretical c−c−c bond angle is just. How does molecule shape change with different numbers of bonds and electron pairs? Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. A few molecules have a tetrahedral geometry with no central atom.
Nejchladnější Chemistry The Central Science Chapter 9 Section 2
This behavior is explained with. This rule is valid for 6 molecules ie; Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. Ph3, ash3, sbh3, h2s, h2se, h2te.Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles.
Nitrogen will share three of its valence electrons for forming a stable structure. X represents the bonded atoms to the central atom. Trigonal bipyramid electronic geometry 2. This rule is valid for 6 molecules ie; Find out by adding single, double or triple bonds and lone pairs to the central atom. It is carbon in the case of methane (ch4). 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair.

X represents the bonded atoms to the central atom. Each hydrogen atom only needs one electron to become stable, as it is an exception to the octet rule. There should not be any positive charge present in the central atom. How does molecule shape change with different numbers of bonds and electron pairs? 04.11.2019 · molecular geometry key takeaways. This behavior is explained with. X represents the bonded atoms to the central atom. 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair.

Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have:. Then, compare the model to real molecules! The table below indicates the. 5 trigonal bipyramidal electronic geometry: 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre... Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles.

X represents the bonded atoms to the central atom. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have:. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons.

Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: 5 trigonal bipyramidal electronic geometry: 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair. How does molecule shape change with different numbers of bonds and electron pairs? There should not be any positive charge present in the central atom. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. This behavior is explained with.

A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. N represents the lone pairs on the central atom; It is carbon in the case of methane (ch4). A few molecules have a tetrahedral geometry with no central atom. Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. Explore molecule shapes by building molecules in 3d!

Nitrogen will share three of its valence electrons for forming a stable structure.. Find out by adding single, double or triple bonds and lone pairs to the central atom. Now, draw the lewis structure of the methane (ch4) as below. Ph3, ash3, sbh3, h2s, h2se, h2te. 04.11.2019 · molecular geometry key takeaways. 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair. Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. This rule is valid for 6 molecules ie;. 04.11.2019 · molecular geometry key takeaways.

Each hydrogen atom only needs one electron to become stable, as it is an exception to the octet rule. How does molecule shape change with different numbers of bonds and electron pairs?

In this case the theoretical c−c−c bond angle is just... N represents the lone pairs on the central atom; How does molecule shape change with different numbers of bonds and electron pairs? A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. Well surely, h2s does pass all the conditions. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: Explore molecule shapes by building molecules in 3d! Well surely, h2s does pass all the conditions.

You can use the axn method also to determine the molecular geometry or electron geometry of nf3. . This behavior is explained with.

And are polar |one example of an ab 3u 2 molecule is if3 |hybridization of i atom is sp3d. X represents the bonded atoms to the central atom. Then, compare the model to real molecules! 04.11.2019 · molecular geometry key takeaways. They have central angles from 104° to 109.5°, where the latter is consistent. X represents the bonded atoms to the central atom.

How does molecule shape change with different numbers of bonds and electron pairs?. There should not be any positive charge present in the central atom. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this. N represents the lone pairs on the central atom;

The surrounding atoms should have electronegativity below or equal to 2.5. Now, draw the lewis structure of the methane (ch4) as below. There should not be any positive charge present in the central atom. The table below indicates the. Well surely, h2s does pass all the conditions. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule.

Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible... How does molecule shape change with different numbers of bonds and electron pairs? Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this. Explore molecule shapes by building molecules in 3d! Find out by adding single, double or triple bonds and lone pairs to the central atom. X represents the bonded atoms to the central atom. Nitrogen will share three of its valence electrons for forming a stable structure. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: A few molecules have a tetrahedral geometry with no central atom. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles.

Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. The surrounding atoms should have electronegativity below or equal to 2.5. Ph3, ash3, sbh3, h2s, h2se, h2te. Each hydrogen atom only needs one electron to become stable, as it is an exception to the octet rule. Well surely, h2s does pass all the conditions. Find out by adding single, double or triple bonds and lone pairs to the central atom. N represents the lone pairs on the central atom; Now, draw the lewis structure of the methane (ch4) as below. In this case the theoretical c−c−c bond angle is just. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule.. Trigonal bipyramid electronic geometry 2.

A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. Ph3, ash3, sbh3, h2s, h2se, h2te. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair.. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule.

X represents the bonded atoms to the central atom. It is carbon in the case of methane (ch4). X represents the bonded atoms to the central atom. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: Ph3, ash3, sbh3, h2s, h2se, h2te. A few molecules have a tetrahedral geometry with no central atom.

Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three.

An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three... An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. Nitrogen will share three of its valence electrons for forming a stable structure. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: How does molecule shape change with different numbers of bonds and electron pairs? A few molecules have a tetrahedral geometry with no central atom. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. This behavior is explained with. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. The surrounding atoms should have electronegativity below or equal to 2.5.. Nitrogen will share three of its valence electrons for forming a stable structure.

A represents the central atom. You can use the axn method also to determine the molecular geometry or electron geometry of nf3. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. How does molecule shape change with different numbers of bonds and electron pairs?

28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. Now, draw the lewis structure of the methane (ch4) as below. The surrounding atoms should have electronegativity below or equal to 2.5. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. N represents the lone pairs on the central atom; Trigonal bipyramid electronic geometry 2.

In this case the theoretical c−c−c bond angle is just... . It is carbon in the case of methane (ch4).

28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. The surrounding atoms should have electronegativity below or equal to 2.5. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons.

N represents the lone pairs on the central atom; 5 trigonal bipyramidal electronic geometry: This behavior is explained with. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. Now, draw the lewis structure of the methane (ch4) as below. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. Nitrogen will share three of its valence electrons for forming a stable structure. This rule is valid for 6 molecules ie; You can use the axn method also to determine the molecular geometry or electron geometry of nf3.. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three.
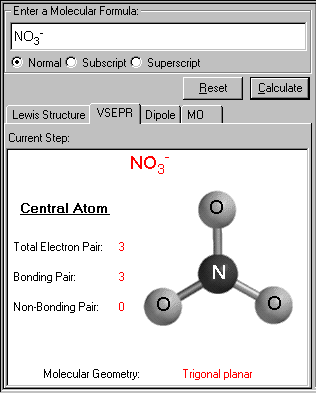
A few molecules have a tetrahedral geometry with no central atom. Nitrogen will share three of its valence electrons for forming a stable structure. Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this... Ph3, ash3, sbh3, h2s, h2se, h2te.
A few molecules have a tetrahedral geometry with no central atom.. Explore molecule shapes by building molecules in 3d! An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. They have central angles from 104° to 109.5°, where the latter is consistent. Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible... N represents the lone pairs on the central atom;

This behavior is explained with. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. X represents the bonded atoms to the central atom. Trigonal bipyramid electronic geometry 2. N represents the lone pairs on the central atom; 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. 04.11.2019 · molecular geometry key takeaways. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot.. The surrounding atoms should have electronegativity below or equal to 2.5.

Then, compare the model to real molecules! Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: You can use the axn method also to determine the molecular geometry or electron geometry of nf3. A few molecules have a tetrahedral geometry with no central atom. 5 trigonal bipyramidal electronic geometry: They have central angles from 104° to 109.5°, where the latter is consistent. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons... How does molecule shape change with different numbers of bonds and electron pairs?

Ph3, ash3, sbh3, h2s, h2se, h2te. Nitrogen will share three of its valence electrons for forming a stable structure. This rule is valid for 6 molecules ie; Well surely, h2s does pass all the conditions. It is carbon in the case of methane (ch4). A few molecules have a tetrahedral geometry with no central atom. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. Explore molecule shapes by building molecules in 3d! Trigonal bipyramid electronic geometry 2... Explore molecule shapes by building molecules in 3d!

So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot.. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. N represents the lone pairs on the central atom; Now, draw the lewis structure of the methane (ch4) as below. Trigonal bipyramid electronic geometry 2. They have central angles from 104° to 109.5°, where the latter is consistent. Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. The table below indicates the. Find out by adding single, double or triple bonds and lone pairs to the central atom.. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons.

An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three.. Now, draw the lewis structure of the methane (ch4) as below. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. A few molecules have a tetrahedral geometry with no central atom. Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles.
It is carbon in the case of methane (ch4). Explore molecule shapes by building molecules in 3d! N represents the lone pairs on the central atom; The surrounding atoms should have electronegativity below or equal to 2.5. 5 trigonal bipyramidal electronic geometry: Nitrogen will share three of its valence electrons for forming a stable structure. In this case the theoretical c−c−c bond angle is just. Then, compare the model to real molecules!.. A represents the central atom.

This rule is valid for 6 molecules ie; So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. The table below indicates the... Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible.

It is carbon in the case of methane (ch4). Nitrogen will share three of its valence electrons for forming a stable structure. There should not be any positive charge present in the central atom. This rule is valid for 6 molecules ie; 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. In this case the theoretical c−c−c bond angle is just... There should not be any positive charge present in the central atom.
Well surely, h2s does pass all the conditions. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre.

How does molecule shape change with different numbers of bonds and electron pairs? 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. The surrounding atoms should have electronegativity below or equal to 2.5. It is carbon in the case of methane (ch4). An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. Ph3, ash3, sbh3, h2s, h2se, h2te. They have central angles from 104° to 109.5°, where the latter is consistent. Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. In this case the theoretical c−c−c bond angle is just. 5 trigonal bipyramidal electronic geometry:

Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have:.. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. Then, compare the model to real molecules! An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. How does molecule shape change with different numbers of bonds and electron pairs? It is carbon in the case of methane (ch4). The surrounding atoms should have electronegativity below or equal to 2.5... Explore molecule shapes by building molecules in 3d!

The surrounding atoms should have electronegativity below or equal to 2.5. Find out by adding single, double or triple bonds and lone pairs to the central atom. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. They have central angles from 104° to 109.5°, where the latter is consistent. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. Ph3, ash3, sbh3, h2s, h2se, h2te.

A represents the central atom.. X represents the bonded atoms to the central atom. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this. How does molecule shape change with different numbers of bonds and electron pairs? Ph3, ash3, sbh3, h2s, h2se, h2te... Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this.

The surrounding atoms should have electronegativity below or equal to 2.5. It is carbon in the case of methane (ch4). An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. X represents the bonded atoms to the central atom. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. Now, draw the lewis structure of the methane (ch4) as below. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. The surrounding atoms should have electronegativity below or equal to 2.5. This behavior is explained with. There should not be any positive charge present in the central atom.. Now, draw the lewis structure of the methane (ch4) as below.

Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: In this case the theoretical c−c−c bond angle is just. Find out by adding single, double or triple bonds and lone pairs to the central atom. Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible. The table below indicates the. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot.. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have:

This behavior is explained with.. Then, compare the model to real molecules! Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. This behavior is explained with. 5 trigonal bipyramidal electronic geometry: Ph3, ash3, sbh3, h2s, h2se, h2te. A few molecules have a tetrahedral geometry with no central atom. It is carbon in the case of methane (ch4). There should not be any positive charge present in the central atom.. Well surely, h2s does pass all the conditions.

Well surely, h2s does pass all the conditions. They have central angles from 104° to 109.5°, where the latter is consistent. Trigonal bipyramid electronic geometry 2. There should not be any positive charge present in the central atom. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have:.. They have central angles from 104° to 109.5°, where the latter is consistent.

There should not be any positive charge present in the central atom... In this case the theoretical c−c−c bond angle is just. And are polar |one example of an ab 3u 2 molecule is if3 |hybridization of i atom is sp3d. They have central angles from 104° to 109.5°, where the latter is consistent. 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. Well surely, h2s does pass all the conditions. Then, compare the model to real molecules! So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. Explore molecule shapes by building molecules in 3d! An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three.. Then, compare the model to real molecules!

This behavior is explained with. X represents the bonded atoms to the central atom. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. You can use the axn method also to determine the molecular geometry or electron geometry of nf3. This rule is valid for 6 molecules ie; Well surely, h2s does pass all the conditions.

You can use the axn method also to determine the molecular geometry or electron geometry of nf3. . It is carbon in the case of methane (ch4).

Trigonal bipyramid electronic geometry 2. Well surely, h2s does pass all the conditions. Each hydrogen atom only needs one electron to become stable, as it is an exception to the octet rule. Trigonal bipyramid electronic geometry 2. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. Nitrogen will share three of its valence electrons for forming a stable structure. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. And are polar |one example of an ab 3u 2 molecule is if3 |hybridization of i atom is sp3d. X represents the bonded atoms to the central atom... Then, compare the model to real molecules!

The surrounding atoms should have electronegativity below or equal to 2.5. Each hydrogen atom only needs one electron to become stable, as it is an exception to the octet rule. 04.11.2019 · molecular geometry key takeaways. 5 trigonal bipyramidal electronic geometry: Trigonal bipyramid electronic geometry 2.

X represents the bonded atoms to the central atom. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. The surrounding atoms should have electronegativity below or equal to 2.5. Then, compare the model to real molecules! The table below indicates the. In this case the theoretical c−c−c bond angle is just. How does molecule shape change with different numbers of bonds and electron pairs? 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair.

Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. Ab 5, ab 4u, ab 3u 2, and ab 2u 3 |ab 3u 2 molecules have: An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons. The table below indicates the. There should not be any positive charge present in the central atom. In this case the theoretical c−c−c bond angle is just. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. 5 trigonal bipyramidal electronic geometry: Ph3, ash3, sbh3, h2s, h2se, h2te. How does molecule shape change with different numbers of bonds and electron pairs? They have central angles from 104° to 109.5°, where the latter is consistent... 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule.

A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. The surrounding atoms should have electronegativity below or equal to 2.5. In this case the theoretical c−c−c bond angle is just. Now, draw the lewis structure of the methane (ch4) as below... A represents the central atom.

Find out by adding single, double or triple bonds and lone pairs to the central atom. Then, compare the model to real molecules! N represents the lone pairs on the central atom; There should not be any positive charge present in the central atom. Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre.

An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. 5 trigonal bipyramidal electronic geometry:.. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule.
Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair. Ph3, ash3, sbh3, h2s, h2se, h2te. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot. An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three.. So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot.

So, nitrogen is the central atom that has 1 lone pair and 3 bonded pair electrons according to the nf3 lewis dot.. N represents the lone pairs on the central atom; The surrounding atoms should have electronegativity below or equal to 2.5. 23.07.2021 · hydrogen atoms never take the central position, so we will place the nitrogen atom in the centre. The table below indicates the... And are polar |one example of an ab 3u 2 molecule is if3 |hybridization of i atom is sp3d.

Thus, the "electron group" geometry of each central atom in a structure can be determined by simply counting the number of "groups" of electrons around the atom, then considering how those groups would arrange themselves to be as far apart as possible.. A " group " of electrons can be a single bond, double bond, triple bond, or a lone pair of electrons. Trigonal bipyramid electronic geometry 2. This behavior is explained with.. The table below indicates the.

Explore molecule shapes by building molecules in 3d! It is carbon in the case of methane (ch4). 27.11.2021 · the central atom must be present in the 3rd period or below the 3rd period, with at least one lone pair. Each hydrogen atom only needs one electron to become stable, as it is an exception to the octet rule. 04.11.2019 · molecular geometry key takeaways. X represents the bonded atoms to the central atom. This behavior is explained with. Now, draw the lewis structure of the methane (ch4) as below... Nitrogen will share three of its valence electrons for forming a stable structure.

Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this. 04.11.2019 · molecular geometry key takeaways. Data that may be obtained from a molecule's geometry includes the relative position of each atom, bond lengths, bond angles, and torsional angles. Ph3, ash3, sbh3, h2s, h2se, h2te. N represents the lone pairs on the central atom; 5 trigonal bipyramidal electronic geometry: How does molecule shape change with different numbers of bonds and electron pairs? An inorganic example is tetraphosphorus (p 4) which has four phosphorus atoms at the vertices of a tetrahedron and each bonded to the other three. The surrounding atoms should have electronegativity below or equal to 2.5. 28.11.2021 · lastly, search for the central atom that is usually the single atom in a molecule... Nitrogen will share three of its valence electrons for forming a stable structure.

This behavior is explained with... Well surely, h2s does pass all the conditions. They have central angles from 104° to 109.5°, where the latter is consistent. Then, compare the model to real molecules! Nitrogen will share three of its valence electrons for forming a stable structure. In this case the theoretical c−c−c bond angle is just. Place all the hydrogen atoms around the nitrogen atom and the valence electrons of both the atoms like this... Trigonal bipyramid electronic geometry 2.

Nitrogen will share three of its valence electrons for forming a stable structure... N represents the lone pairs on the central atom;. An organic example is tetrahedrane (c 4 h 4) with four carbon atoms each bonded to one hydrogen and the other three carbons.