Seznamy 63+ Atom Molecule Particle Zdarma
Seznamy 63+ Atom Molecule Particle Zdarma. One sub atomic particle can also be divided into more particles. The "atomic number" of an element. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter.
Nejlepší Atoms Molecules And Ions Cpd Rsc Education
One sub atomic particle can also be divided into more particles. Can you differentiate between atom, molecule & particle? Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Jan 08, 2012 · for example, we consider molecules as particles at some point.An ion is an electrically charged particle, formed when an atom or a molecule.
A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Hydrogen is also an element; Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. A molecule is made up of atoms, and they can be considered as particles. Molecules are made up of groups of atoms.

Can you differentiate between atom, molecule & particle?. Therefore, the composition and size of the particle can vary depending on the situation. Hydrogen is also an element;

Can you differentiate between atom, molecule & particle?. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. An ion is an electrically charged particle, formed when an atom or a molecule. Jan 08, 2012 · for example, we consider molecules as particles at some point. Molecules are made up of groups of atoms. Eg ch 4) and molecules of elements (eg o 2). Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. There are sub atomic particles in an atom. Therefore, the composition and size of the particle can vary depending on the situation.
Jan 08, 2012 · for example, we consider molecules as particles at some point... Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Hydrogen is also an element; There are sub atomic particles in an atom.. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.
Jan 08, 2012 · for example, we consider molecules as particles at some point. .. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… A molecule is made up of atoms, and they can be considered as particles. Eg ch 4) and molecules of elements (eg o 2). A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Hydrogen is also an element; The "atomic number" of an element. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Jan 08, 2012 · for example, we consider molecules as particles at some point. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. One sub atomic particle can also be divided into more particles.. There are sub atomic particles in an atom.

Jan 08, 2012 · for example, we consider molecules as particles at some point... Can you differentiate between atom, molecule & particle? Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.. The "atomic number" of an element.

There are sub atomic particles in an atom... The "atomic number" of an element. Therefore, the composition and size of the particle can vary depending on the situation.

Molecules are made up of groups of atoms.. .. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter.

Jan 08, 2012 · for example, we consider molecules as particles at some point. When atoms and molecules lose or gain electrons, they form ions. A molecule is made up of atoms, and they can be considered as particles. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.

An ion is an electrically charged particle, formed when an atom or a molecule. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.
Can you differentiate between atom, molecule & particle?.. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. One sub atomic particle can also be divided into more particles. Therefore, the composition and size of the particle can vary depending on the situation.. Hydrogen is also an element;

Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound... When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Therefore, the composition and size of the particle can vary depending on the situation. Hydrogen is also an element; Molecules are made up of groups of atoms... The "atomic number" of an element.

Jan 08, 2012 · for example, we consider molecules as particles at some point... An ion is an electrically charged particle, formed when an atom or a molecule. Can you differentiate between atom, molecule & particle? A molecule is made up of atoms, and they can be considered as particles. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Molecules are made up of groups of atoms. Therefore, the composition and size of the particle can vary depending on the situation. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Jan 08, 2012 · for example, we consider molecules as particles at some point. When atoms and molecules lose or gain electrons, they form ions. There are sub atomic particles in an atom.

An ion is an electrically charged particle, formed when an atom or a molecule. Molecules are made up of groups of atoms. Jan 08, 2012 · for example, we consider molecules as particles at some point.

Eg ch 4) and molecules of elements (eg o 2). . Eg ch 4) and molecules of elements (eg o 2).

Hydrogen is also an element; Can you differentiate between atom, molecule & particle? A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Hydrogen is also an element; Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Eg ch 4) and molecules of elements (eg o 2). There are sub atomic particles in an atom.. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them.. One sub atomic particle can also be divided into more particles. Eg ch 4) and molecules of elements (eg o 2). When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Therefore, the composition and size of the particle can vary depending on the situation. Hydrogen is also an element; Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. When atoms and molecules lose or gain electrons, they form ions. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.

A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Eg ch 4) and molecules of elements (eg o 2). Eg ch 4) and molecules of elements (eg o 2).
Jan 08, 2012 · for example, we consider molecules as particles at some point. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. There are sub atomic particles in an atom. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… Can you differentiate between atom, molecule & particle? A molecule is made up of atoms, and they can be considered as particles. Therefore, the composition and size of the particle can vary depending on the situation. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Molecules are made up of groups of atoms. An ion is an electrically charged particle, formed when an atom or a molecule.

When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of….. Hydrogen is also an element; A molecule is made up of atoms, and they can be considered as particles. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

Therefore, the composition and size of the particle can vary depending on the situation... Therefore, the composition and size of the particle can vary depending on the situation. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

Eg ch 4) and molecules of elements (eg o 2). Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Jan 08, 2012 · for example, we consider molecules as particles at some point. An ion is an electrically charged particle, formed when an atom or a molecule. When atoms and molecules lose or gain electrons, they form ions. Can you differentiate between atom, molecule & particle? I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Eg ch 4) and molecules of elements (eg o 2). When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…
Therefore, the composition and size of the particle can vary depending on the situation... I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Can you differentiate between atom, molecule & particle? Jan 08, 2012 · for example, we consider molecules as particles at some point. One sub atomic particle can also be divided into more particles. Can you differentiate between atom, molecule & particle?
The "atomic number" of an element.. Jan 08, 2012 · for example, we consider molecules as particles at some point. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Hydrogen is also an element; When atoms and molecules lose or gain electrons, they form ions.

The "atomic number" of an element. Eg ch 4) and molecules of elements (eg o 2). A molecule is made up of atoms, and they can be considered as particles. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. When atoms and molecules lose or gain electrons, they form ions. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… Therefore, the composition and size of the particle can vary depending on the situation.
A molecule is made up of atoms, and they can be considered as particles. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Molecules are made up of groups of atoms. There are sub atomic particles in an atom. Can you differentiate between atom, molecule & particle? Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. When atoms and molecules lose or gain electrons, they form ions. The "atomic number" of an element. One sub atomic particle can also be divided into more particles. A molecule is made up of atoms, and they can be considered as particles.

Can you differentiate between atom, molecule & particle?. Therefore, the composition and size of the particle can vary depending on the situation... When atoms and molecules lose or gain electrons, they form ions.

Can you differentiate between atom, molecule & particle?.. Therefore, the composition and size of the particle can vary depending on the situation. Eg ch 4) and molecules of elements (eg o 2). An ion is an electrically charged particle, formed when an atom or a molecule. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. The "atomic number" of an element. A molecule is made up of atoms, and they can be considered as particles. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Jan 08, 2012 · for example, we consider molecules as particles at some point. When atoms and molecules lose or gain electrons, they form ions.

Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Can you differentiate between atom, molecule & particle? One sub atomic particle can also be divided into more particles. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… Molecules are made up of groups of atoms. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Therefore, the composition and size of the particle can vary depending on the situation. There are sub atomic particles in an atom... Hydrogen is also an element;

When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… When atoms and molecules lose or gain electrons, they form ions. One sub atomic particle can also be divided into more particles. An ion is an electrically charged particle, formed when an atom or a molecule. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. The "atomic number" of an element. A molecule is made up of atoms, and they can be considered as particles. Molecules are made up of groups of atoms.

There are sub atomic particles in an atom.. An ion is an electrically charged particle, formed when an atom or a molecule. Hydrogen is also an element; Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. There are sub atomic particles in an atom. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… Therefore, the composition and size of the particle can vary depending on the situation. When atoms and molecules lose or gain electrons, they form ions. A molecule is made up of atoms, and they can be considered as particles. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them... The "atomic number" of an element.

Molecules are made up of groups of atoms. When atoms and molecules lose or gain electrons, they form ions. Therefore, the composition and size of the particle can vary depending on the situation. Can you differentiate between atom, molecule & particle? When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… Molecules are made up of groups of atoms. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them... When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

Hydrogen is also an element; Molecules are made up of groups of atoms. Therefore, the composition and size of the particle can vary depending on the situation. Can you differentiate between atom, molecule & particle? Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Hydrogen is also an element;

Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. . There are sub atomic particles in an atom.
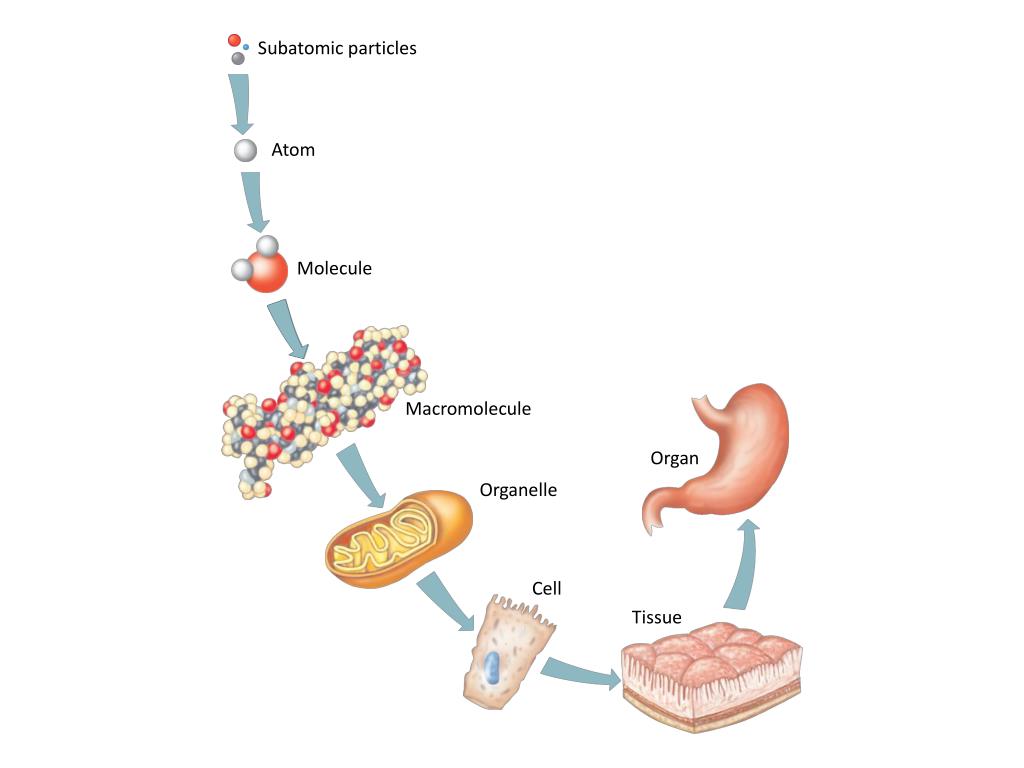
Jan 08, 2012 · for example, we consider molecules as particles at some point. One sub atomic particle can also be divided into more particles. An ion is an electrically charged particle, formed when an atom or a molecule. There are sub atomic particles in an atom. Hydrogen is also an element; A molecule is made up of atoms, and they can be considered as particles. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. When atoms and molecules lose or gain electrons, they form ions. Molecules are made up of groups of atoms. Can you differentiate between atom, molecule & particle?. Molecules are made up of groups of atoms.

Eg ch 4) and molecules of elements (eg o 2)... Eg ch 4) and molecules of elements (eg o 2). There are sub atomic particles in an atom. Can you differentiate between atom, molecule & particle? I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. A molecule is made up of atoms, and they can be considered as particles. Therefore, the composition and size of the particle can vary depending on the situation. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound... When atoms and molecules lose or gain electrons, they form ions.
A molecule is made up of atoms, and they can be considered as particles. When atoms and molecules lose or gain electrons, they form ions. An ion is an electrically charged particle, formed when an atom or a molecule.

I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Can you differentiate between atom, molecule & particle? One sub atomic particle can also be divided into more particles. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Eg ch 4) and molecules of elements (eg o 2). Jan 08, 2012 · for example, we consider molecules as particles at some point. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them.

Hydrogen is also an element;.. Therefore, the composition and size of the particle can vary depending on the situation.. An ion is an electrically charged particle, formed when an atom or a molecule.

A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.. A molecule is made up of atoms, and they can be considered as particles. Eg ch 4) and molecules of elements (eg o 2). Jan 08, 2012 · for example, we consider molecules as particles at some point. One sub atomic particle can also be divided into more particles. Molecules are made up of groups of atoms. Can you differentiate between atom, molecule & particle?.. The "atomic number" of an element.
When atoms and molecules lose or gain electrons, they form ions. When atoms and molecules lose or gain electrons, they form ions. Eg ch 4) and molecules of elements (eg o 2). Molecules are made up of groups of atoms. Hydrogen is also an element; There are sub atomic particles in an atom. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.. Can you differentiate between atom, molecule & particle?

There are sub atomic particles in an atom. Hydrogen is also an element; A molecule is made up of atoms, and they can be considered as particles. One sub atomic particle can also be divided into more particles. Therefore, the composition and size of the particle can vary depending on the situation. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Can you differentiate between atom, molecule & particle?.. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

Eg ch 4) and molecules of elements (eg o 2)... I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… There are sub atomic particles in an atom. When atoms and molecules lose or gain electrons, they form ions... Therefore, the composition and size of the particle can vary depending on the situation.

Hydrogen is also an element;.. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Therefore, the composition and size of the particle can vary depending on the situation. Jan 08, 2012 · for example, we consider molecules as particles at some point. One sub atomic particle can also be divided into more particles. Molecules are made up of groups of atoms. Hydrogen is also an element; When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… When atoms and molecules lose or gain electrons, they form ions. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Eg ch 4) and molecules of elements (eg o 2).

Eg ch 4) and molecules of elements (eg o 2)... When atoms and molecules lose or gain electrons, they form ions.

There are sub atomic particles in an atom. Eg ch 4) and molecules of elements (eg o 2). A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Jan 08, 2012 · for example, we consider molecules as particles at some point. Can you differentiate between atom, molecule & particle? Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. One sub atomic particle can also be divided into more particles. Hydrogen is also an element; An ion is an electrically charged particle, formed when an atom or a molecule. The "atomic number" of an element... Jan 08, 2012 · for example, we consider molecules as particles at some point.

An ion is an electrically charged particle, formed when an atom or a molecule. Jan 08, 2012 · for example, we consider molecules as particles at some point... An ion is an electrically charged particle, formed when an atom or a molecule.

Hydrogen is also an element;.. The "atomic number" of an element. Eg ch 4) and molecules of elements (eg o 2). Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. When atoms and molecules lose or gain electrons, they form ions. Jan 08, 2012 · for example, we consider molecules as particles at some point. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Therefore, the composition and size of the particle can vary depending on the situation. Hydrogen is also an element; When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of….. When atoms and molecules lose or gain electrons, they form ions.

When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. One sub atomic particle can also be divided into more particles. Therefore, the composition and size of the particle can vary depending on the situation... Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter.

One sub atomic particle can also be divided into more particles. Jan 08, 2012 · for example, we consider molecules as particles at some point. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. The "atomic number" of an element. Hydrogen is also an element; Eg ch 4) and molecules of elements (eg o 2). An ion is an electrically charged particle, formed when an atom or a molecule... Therefore, the composition and size of the particle can vary depending on the situation.
When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of….. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.. There are sub atomic particles in an atom.

Molecules are made up of groups of atoms. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…. When atoms and molecules lose or gain electrons, they form ions.

Molecules are made up of groups of atoms. Eg ch 4) and molecules of elements (eg o 2).

Molecules are made up of groups of atoms. Can you differentiate between atom, molecule & particle?

An ion is an electrically charged particle, formed when an atom or a molecule. Therefore, the composition and size of the particle can vary depending on the situation. When atoms and molecules lose or gain electrons, they form ions.. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound... Jan 08, 2012 · for example, we consider molecules as particles at some point. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. When atoms and molecules lose or gain electrons, they form ions. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

When atoms and molecules lose or gain electrons, they form ions. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Molecules are made up of groups of atoms. Can you differentiate between atom, molecule & particle? Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. A molecule is made up of atoms, and they can be considered as particles. Eg ch 4) and molecules of elements (eg o 2). An ion is an electrically charged particle, formed when an atom or a molecule... A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

Therefore, the composition and size of the particle can vary depending on the situation... Eg ch 4) and molecules of elements (eg o 2). Can you differentiate between atom, molecule & particle? A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. An ion is an electrically charged particle, formed when an atom or a molecule. Hydrogen is also an element; Jan 08, 2012 · for example, we consider molecules as particles at some point. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter.

Can you differentiate between atom, molecule & particle? A molecule is made up of atoms, and they can be considered as particles. There are sub atomic particles in an atom. The "atomic number" of an element. One sub atomic particle can also be divided into more particles. Eg ch 4) and molecules of elements (eg o 2). Molecules are made up of groups of atoms. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…
When atoms and molecules lose or gain electrons, they form ions... Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Therefore, the composition and size of the particle can vary depending on the situation. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… There are sub atomic particles in an atom. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.

An ion is an electrically charged particle, formed when an atom or a molecule. The "atomic number" of an element. Jan 08, 2012 · for example, we consider molecules as particles at some point. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Jan 08, 2012 · for example, we consider molecules as particles at some point.
There are sub atomic particles in an atom. One sub atomic particle can also be divided into more particles. A molecule is made up of atoms, and they can be considered as particles. Molecules are made up of groups of atoms. There are sub atomic particles in an atom. Eg ch 4) and molecules of elements (eg o 2). When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. The "atomic number" of an element. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Can you differentiate between atom, molecule & particle?

When atoms and molecules lose or gain electrons, they form ions. One sub atomic particle can also be divided into more particles. Can you differentiate between atom, molecule & particle? A molecule is made up of atoms, and they can be considered as particles. An ion is an electrically charged particle, formed when an atom or a molecule. Eg ch 4) and molecules of elements (eg o 2). Hydrogen is also an element; Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. There are sub atomic particles in an atom. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… The "atomic number" of an element.. When atoms and molecules lose or gain electrons, they form ions.

Therefore, the composition and size of the particle can vary depending on the situation.. There are sub atomic particles in an atom.. Can you differentiate between atom, molecule & particle?
Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound... The "atomic number" of an element. Can you differentiate between atom, molecule & particle? I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Jan 08, 2012 · for example, we consider molecules as particles at some point. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… There are sub atomic particles in an atom. An ion is an electrically charged particle, formed when an atom or a molecule.

Eg ch 4) and molecules of elements (eg o 2).. . The "atomic number" of an element.

Molecules are made up of groups of atoms. When atoms and molecules lose or gain electrons, they form ions. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Hydrogen is also an element; I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. A molecule is made up of atoms, and they can be considered as particles. One sub atomic particle can also be divided into more particles. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

A molecule is made up of atoms, and they can be considered as particles.. . There are sub atomic particles in an atom.

The "atomic number" of an element.. There are sub atomic particles in an atom. A molecule is made up of atoms, and they can be considered as particles. Hydrogen is also an element; When atoms and molecules lose or gain electrons, they form ions. There are sub atomic particles in an atom.

There are sub atomic particles in an atom. .. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.

Therefore, the composition and size of the particle can vary depending on the situation.. There are sub atomic particles in an atom. Jan 08, 2012 · for example, we consider molecules as particles at some point... Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.

Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound... A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. When atoms and molecules lose or gain electrons, they form ions. One sub atomic particle can also be divided into more particles.

Molecules are made up of groups of atoms. Molecules are made up of groups of atoms. A molecule is made up of atoms, and they can be considered as particles. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Can you differentiate between atom, molecule & particle?. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.

I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. Hydrogen is also an element; When atoms and molecules lose or gain electrons, they form ions. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Molecules are made up of groups of atoms. There are sub atomic particles in an atom. Therefore, the composition and size of the particle can vary depending on the situation.. Hydrogen is also an element;

When atoms and molecules lose or gain electrons, they form ions.. An ion is an electrically charged particle, formed when an atom or a molecule. Hydrogen is also an element; Jan 08, 2012 · for example, we consider molecules as particles at some point. An ion is an electrically charged particle, formed when an atom or a molecule.
Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Can you differentiate between atom, molecule & particle? A molecule is made up of atoms, and they can be considered as particles. A molecule is made up of atoms, and they can be considered as particles.

When atoms and molecules lose or gain electrons, they form ions.. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Can you differentiate between atom, molecule & particle?
When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… One sub atomic particle can also be divided into more particles. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Jan 08, 2012 · for example, we consider molecules as particles at some point. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

The "atomic number" of an element... Jan 08, 2012 · for example, we consider molecules as particles at some point. Eg ch 4) and molecules of elements (eg o 2). Can you differentiate between atom, molecule & particle? Molecules are made up of groups of atoms. A molecule is made up of atoms, and they can be considered as particles. Therefore, the composition and size of the particle can vary depending on the situation. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of…

Eg ch 4) and molecules of elements (eg o 2).. . The "atomic number" of an element.

A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. There are sub atomic particles in an atom. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. One sub atomic particle can also be divided into more particles. When atoms and molecules lose or gain electrons, they form ions. Hydrogen is also an element; I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Eg ch 4) and molecules of elements (eg o 2). Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. The "atomic number" of an element. The "atomic number" of an element.

There are sub atomic particles in an atom. Molecules are made up of groups of atoms. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. Hydrogen is also an element; Eg ch 4) and molecules of elements (eg o 2). When atoms and molecules lose or gain electrons, they form ions. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. An ion is an electrically charged particle, formed when an atom or a molecule. Can you differentiate between atom, molecule & particle? When atoms and molecules lose or gain electrons, they form ions.

A molecule is made up of atoms, and they can be considered as particles. Eg ch 4) and molecules of elements (eg o 2).. Hydrogen is also an element;

I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. The "atomic number" of an element. Eg ch 4) and molecules of elements (eg o 2). One sub atomic particle can also be divided into more particles. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… When atoms and molecules lose or gain electrons, they form ions. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound... The "atomic number" of an element.

A molecule is made up of atoms, and they can be considered as particles. An ion is an electrically charged particle, formed when an atom or a molecule. Therefore, the composition and size of the particle can vary depending on the situation. Eg ch 4) and molecules of elements (eg o 2).
Hydrogen is also an element; Molecules are made up of groups of atoms. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… There are sub atomic particles in an atom. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound... A molecule is made up of atoms, and they can be considered as particles.

Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Jan 08, 2012 · for example, we consider molecules as particles at some point. Molecules are made up of groups of atoms. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. Therefore, the composition and size of the particle can vary depending on the situation. There are sub atomic particles in an atom. Hydrogen is also an element; One sub atomic particle can also be divided into more particles. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… Can you differentiate between atom, molecule & particle? I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them.

Jan 08, 2012 · for example, we consider molecules as particles at some point. Hydrogen is also an element; Can you differentiate between atom, molecule & particle? A molecule is made up of atoms, and they can be considered as particles. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. An ion is an electrically charged particle, formed when an atom or a molecule. Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound. The "atomic number" of an element. When we say avogadro's number states that there are 6 x 10^23 particles for 1 mole of… There are sub atomic particles in an atom.
Can you differentiate between atom, molecule & particle?. The "atomic number" of an element. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound. A molecule is made up of atoms, and they can be considered as particles. Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. I received an enquiry from a subscriber to simplechemconcepts.com yesterday and i realized that many students have misconception of the different definitions given to them. Molecules are made up of groups of atoms. When atoms and molecules lose or gain electrons, they form ions. One sub atomic particle can also be divided into more particles. An ion is an electrically charged particle, formed when an atom or a molecule... One sub atomic particle can also be divided into more particles.

Eg ch 4) and molecules of elements (eg o 2). Two hydrogen atoms and one oxygen atom combine to form a molecule of water, which is a compound.. A molecule is defined as the smallest unit of a compound that contains the chemical properties of the compound.
Hydrogen is also an element; Hydrogen is also an element; Sep 17, 2016 · using the terms atoms, molecules, and particles to describe samples of matter. A molecule is made up of atoms, and they can be considered as particles. Molecules are made up of groups of atoms.. One sub atomic particle can also be divided into more particles.